Heat is a form of energy that is transferred from one body to another due to a difference in temperature. The study of quantity of heat and heat capacity helps us understand how substances absorb, store, and transfer thermal energy. Quantity of heat refers to the amount of thermal energy gained or lost by a substance, while heat capacity is the measure of the amount of heat required to raise the temperature of a body by one degree Celsius or one Kelvin. These concepts are important in explaining everyday phenomena such as heating water, cooking food, and the functioning of heating systems. Understanding quantity of heat and capacity provides a foundation for studying thermal physics and helps in analyzing how different materials respond to heating and cooling processes.
Heat is a form of energy that flows from a region of higher temperature to a region of lower temperature.
When heat is supplied to a substance, two things may happen:
- The temperature of the substance increases
- The substance changes its state (for example, melting or boiling)
Understanding how heat affects matter is important in physics, engineering, and everyday life.
Heat and Temperature Change
The rise in temperature of a substance depends on:
- The mass of the substance
- The type of material
- The amount of heat supplied
This explains why water heats more slowly than metals.
Heat Capacity
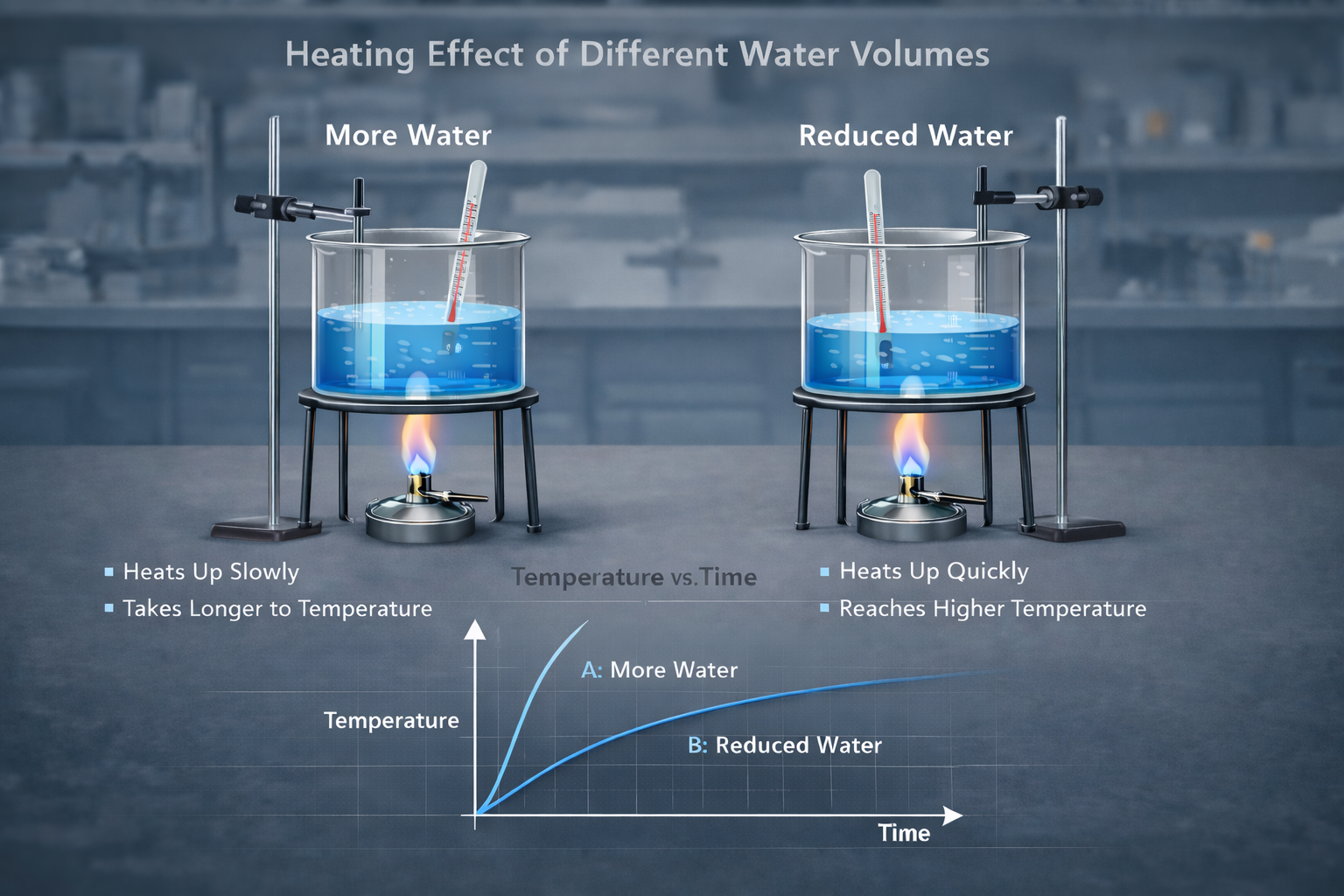
The figure below illustrates comparing of different capacity of water being heated for sometimes by a bunsen burner. The beakers are identical and the graph shows how their temperature changes with time.

Definition:
Heat capacity is the amount of heat required to raise the temperature of a body by 1°C (or 1 K).
Formula:
Q = Cθ
Where:
Q = heat energy (J)
C = heat capacity (J K⁻¹)
θ = temperature change (°C or K)
Specific Heat Capacity
Definition:
Specific heat capacity is the amount of heat required to raise the temperature of 1 kg of a substance by 1 K.
Formula:
Q = mcθ
Where:
m = mass (kg)
c = specific heat capacity (J kg⁻¹ K⁻¹)
θ = temperature change
Key Concept
Different materials respond differently to heat:
- Water has a high specific heat capacity → heats slowly
- Metals have low specific heat capacity → heat quickly
Relationship Between Heat Capacity and Specific Heat Capacity
C = mc
This means total heat capacity depends on both:
- Mass
- Type of material
Heating Experiment
Aim:
To observe how heat affects temperature change in water.
Procedure:
- Measure a fixed volume of water
- Record its initial temperature
- Heat the water
- Record time taken to reach a higher temperature
- Repeat using different volumes
Observation:
- Larger volume → heats more slowly
- Smaller volume → heats faster
Conclusion:
The quantity of heat required depends on the mass of the substance.
Worked Examples
Example 1
Heat capacity = 460 J K⁻¹
Temperature change = 45°C − 15°C = 30°C
Q = Cθ
Q = 460 × 30
Q = 13,800 J
Example 2
Power = 50 W
Time = 9 minutes = 540 s
Q = Pt
Q = 50 × 540
Q = 27,000 J
Example 3
Mass = 60 g = 0.06 kg
Specific heat capacity = 390 J kg⁻¹ K⁻¹
Temperature change = 50°C
Q = mcθ
Q = 0.06 × 390 × 50
Q = 1170 J
Table of Specific Heat Capacities
| Substance | Value (J kg⁻¹ K⁻¹) |
|---|---|
| Water | 4200 |
| Alcohol | 2400 |
| Kerosene | 2200 |
| Ice | 2100 |
| Aluminium | 900 |
| Copper | 390 |
| Lead | 130 |